By Ian O’Neill, Quadram Institute Bioscience, Norwich, UK

Over the past two decades the research into the ‘microbiome’ has increased substantially and has revealed that there are complex ecosystems of microbes (bacteria, yeast, fungi and viruses) living on or within us from birth. Each surface of the body that is exposed to the outside world (lungs, gut, skin etc) contains its own profile of microbes unique to each person and these microbes contribute to our health and shape our immune system as we grow. The gut contains the largest concentration of bacteria in the body but surprisingly we only have a single epithelial cell layer that prevents the bacteria from entering our bloodstream and causing fatal blood infections. Even more surprising is that while these cells keep the bacteria out they simultaneously allow water and nutrients from food in. This selective barrier works, in part, due to a continuous belt of proteins called tight junctions that keep neighbouring epithelial cells connected and the barrier intact. In diseases like inflammatory bowel disease (IBD), these tight junctions are disrupted leaving the barrier compromised and allowing bacteria and other material to pass from the intestines into the underlying bloodstream; this phenomenon is sometimes referred to as ‘leaky gut’. The immune system is quick to identify the bacteria that cross the barrier and cells such as dendritic cells and macrophages that patrol the gut engulf and kill the bacteria. This immune response is an inflammatory process and leads to the release of pro-inflammatory cytokines, chemical signals that communicate the presence of a bacteria and cause inflammation and swelling of the the gut wall. This further disrupts the barrier leading to more movement of bacteria across it and thus more inflammation. In IBD, where there is a damaged barrier and excessive inflammation, it is important to reduce the inflammation, as well as repair the barrier function.
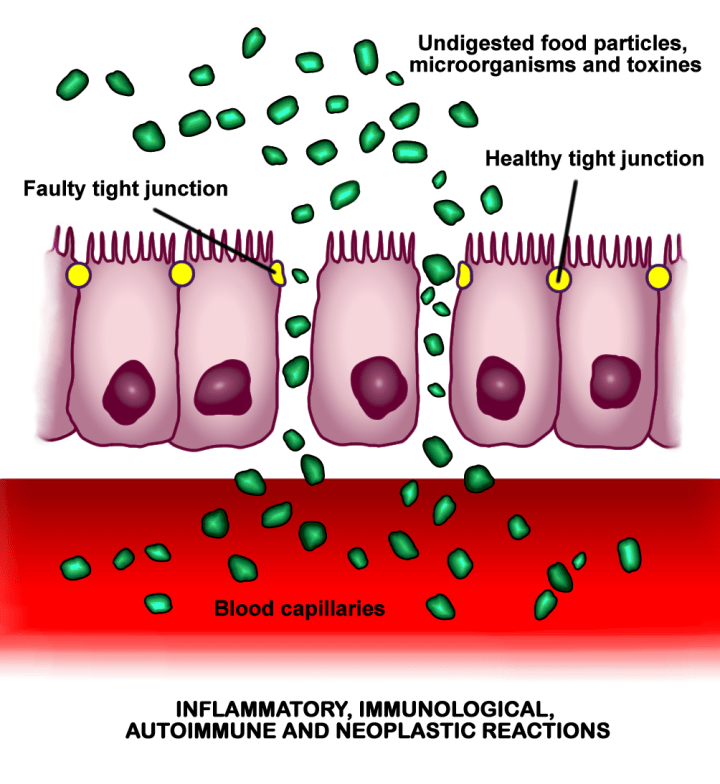
The exact cause of IBD is not known but it is thought to be a combination of inherited genetic factors and a change in the composition of microbes in gut resulting in an increase in ‘bad’ bacteria and a decrease in ‘good’ bacteria. One ‘good’ bacteria that decreases in IBD is Bifidobacterium, a species of bacteria that are commonly used as probiotics – bacteria that are known to promote health. You can typically find them on the shelves in our local shop in in the form of a yogurt drink. Since Bifidobacterium is decreased in IBD there has been some clinical trials where patients are given a supplement of Bifidobacterium to help improve their symptoms. Some of these trials had positive results although more basic research is required to better understand how these bacteria are having a beneficial effect. Some studies have indicated that the beneficial effect may be due to strengthening of tight junctions (through expression of tight junction protein genes). In IBD, there is also excessive shedding of the cells lining the gut that also contributes to a disrupted barrier, but recently we showed that treatment with bifidobacteria reduces this shedding. These studies show great promise for treating patients with gastrointestinal disease with probiotic therapies.
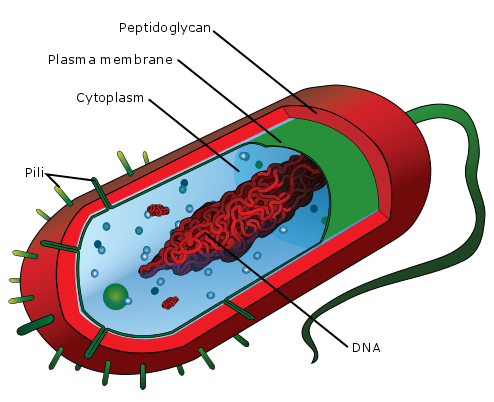
Whilst, there is some research to show how epithelial cells in the gut respond to bifidobacteria, there is less information on what exactly the bacteria are doing to exert this effect. Bacterial cells are structurally complex, however, they fundamentally contain the same components as most other living cells. They have an outer layer which protects the inner ‘soup’ (the cytoplasm) that contains all the components (genes, proteins, enzymes etc.) required for life and replication. The outer layer of bifidobacteria is made up of peptidoglycan, a mesh structured polymer of sugars and amino acids that surrounds the cell. Bacteria have protein based appendages called pili or fimbriae that are attached to their outer layer and are used, in many cases, to attach to the intestines during colonisation. Bifidobacterium possess several different types of pili. One pili called a Type IVb pili has been visualised on a strain called Bifidobacterium breve with electron microscopy, and we know that this pili is required to for the bacteria to live and survive in the gut.
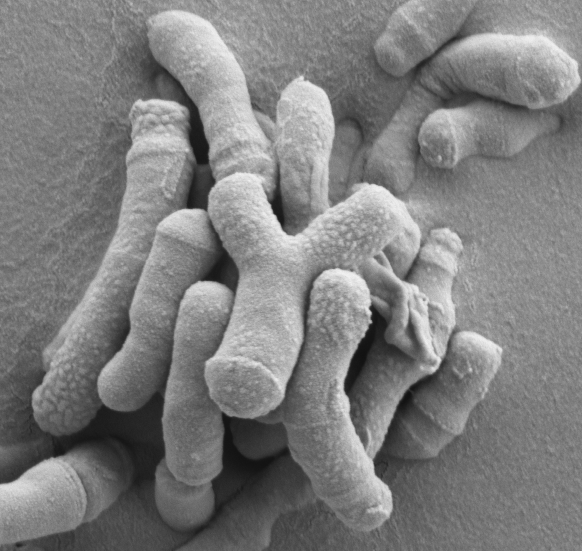
Similarly, sortase dependent pili have been visualised by microscopy and have been suggested to be important in adhesion of bifidobacteria to the epithelial cells that line the gut. Some strains of bifidobacteria also produce a polymer called exopolysaccharide (EPS) that is made up many sugar molecules joined together and in some strains of bifidobacteria EPS remains attached to the surface of the bacteria forming a distinct halo around the cell when viewed with an electron microscope. EPS may play an important role in the beneficial effect bifidobacteria have in the treatment of IBD. Our lab group has recently shown that EPS is important in preventing shedding of the epithelial cells of the gut that occurs in UC. To build on this discovery, I am currently researching if there are more structural or genetic components of Bifidobacterium that contribute to their beneficial effects. With this information, we hope to be able to identify new strains of bifidobacteria that better promote intestinal barrier function that can be used for treatment of IBD.
My work is generously funded by Crohn’s and Colitis UK, UK’s leading charity in the battle against Crohn’s Disease and Ulcerative Colitis; find out more at https://www.crohnsandcolitis.org.uk/. To find out more about the cutting-edge research we do in the Hall lab at the Quadram Institute Bioscience visit our website.
This post is the third in our microbiome series. The first, Microbiome manipulation – full steam ahead? by Tilly Potter, was published in April. The second, Phyte club: inside the plant microbiome, by Gareth Raynes, was published earlier this month. If you are interested in reading more on this topic, check out the April issue of The Biochemist magazine, which focuses on The Microbiome.


One thought on “Lending a helping hand: Can probiotics treat inflammatory bowel disease?”