By David Hornby, University of Sheffield, UK
 Forty years ago this month I sat in my first lecture, an intensive introduction to ‘University Chemistry’. This was partially tempered with an Integrated Biology course on Darwinian evolution. Maybe this ‘christening by fire’ gave me the strong view that all biochemists should be equally grounded in both of these subjects. I also remember being told at school that Darwin’s ideas were all about chance and selection, while Lamarck’s theory centred on inter-generational adaptation. Who hasn’t been told about Lamarck’s giraffes!
Forty years ago this month I sat in my first lecture, an intensive introduction to ‘University Chemistry’. This was partially tempered with an Integrated Biology course on Darwinian evolution. Maybe this ‘christening by fire’ gave me the strong view that all biochemists should be equally grounded in both of these subjects. I also remember being told at school that Darwin’s ideas were all about chance and selection, while Lamarck’s theory centred on inter-generational adaptation. Who hasn’t been told about Lamarck’s giraffes!
Evolving views
Such Victorian imagery is as familiar today as it was then, as the debate over evolution continues as fervently as ever. By comparison, you would be hard pressed to find many popular headlines challenging the fundamental aspects of modern chemistry! Most of the public concern over chemistry in Darwin’s day, related to pollution and public safety. So I shall leave chemistry for now and concentrate on the fundamental mechanisms that underpin evolution and its connection with the topic of this piece: epigenetics.
 Jean-Baptiste Lamarck published his ideas on evolution in 1809, fifty years before Darwin and sixty years before Dmitri Mendeleev’s Periodic Table. Later, during the 1950s, the great French duo of Jacob and Monod, were formulating their model for gene regulation, ultimately recognised by the award of the Nobel Prize in 1965. A little earlier, the embryologist Conrad Waddington and the mathematician (or code-breaker), Alan Turing independently wrestled with ‘programmed’ differential gene expression during embryonic development. So when the Pulitzer Prize winning clinical scientist, Siddhartha Mukherjee published an article in The New Yorker, in Spring of 2016, bringing Lamarck’s world view firmly back into the frame, all (scientific) hell broke loose! It seemed to some that the slumbering giant of Lamarck was about to gain a new audience.
Jean-Baptiste Lamarck published his ideas on evolution in 1809, fifty years before Darwin and sixty years before Dmitri Mendeleev’s Periodic Table. Later, during the 1950s, the great French duo of Jacob and Monod, were formulating their model for gene regulation, ultimately recognised by the award of the Nobel Prize in 1965. A little earlier, the embryologist Conrad Waddington and the mathematician (or code-breaker), Alan Turing independently wrestled with ‘programmed’ differential gene expression during embryonic development. So when the Pulitzer Prize winning clinical scientist, Siddhartha Mukherjee published an article in The New Yorker, in Spring of 2016, bringing Lamarck’s world view firmly back into the frame, all (scientific) hell broke loose! It seemed to some that the slumbering giant of Lamarck was about to gain a new audience.
The birth of modern epigenetics
 The developments in molecular cloning during the 1970s and 1980s, provided molecular biologists with the tools they needed to begin to define the molecular players and pathways underpinning gene regulation. (As a primer, I can strongly recommend the elegantly written book “A Genetic Switch” by one of the early pioneers, Mark Ptashne). Fundamental to these ideas, is the connection between the specific recognition of a gene (or group of genes) by transcription factors, which are usually polypeptides, but can also be RNA molecules.
The developments in molecular cloning during the 1970s and 1980s, provided molecular biologists with the tools they needed to begin to define the molecular players and pathways underpinning gene regulation. (As a primer, I can strongly recommend the elegantly written book “A Genetic Switch” by one of the early pioneers, Mark Ptashne). Fundamental to these ideas, is the connection between the specific recognition of a gene (or group of genes) by transcription factors, which are usually polypeptides, but can also be RNA molecules.
Elsewhere, a small number of laboratories were pondering the genetics and biochemistry of restriction and modification. It soon became clear that the methylation of ‘specific’ DNA sequences, was an integral part of this phenomenon. And so when, in the late 1980s, Timothy Bestor then at MIT, obtained the nucleotide sequence of the first vertebrate DNA methyltransferase (showing strong similarities to bacterial enzymes of this class): a seductive connection between bacterial genetics and vertebrate development was established, for many.
The next problem facing the epigenetics community, was to explain how the differential methylation of a two nucleotide motif, the CpG dinucleotide, which occurs on average every 16 nucleotides in any given genome.
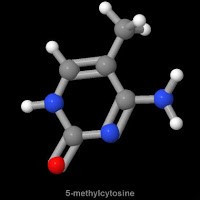 Stepping back in time for a moment, the suggestion that some genomic nucleotides may be modified was first suggested at the close of the nineteenth century, but was only formally confirmed as 5-methylcytosine by the elegant work of Rollin Hotchkiss, that paralleled the work of Erwin Chargaff on genomic DNA, a few years before Watson and Crick’s ground-breaking publication of the double helical nature of DNA.
Stepping back in time for a moment, the suggestion that some genomic nucleotides may be modified was first suggested at the close of the nineteenth century, but was only formally confirmed as 5-methylcytosine by the elegant work of Rollin Hotchkiss, that paralleled the work of Erwin Chargaff on genomic DNA, a few years before Watson and Crick’s ground-breaking publication of the double helical nature of DNA.
However, since the basic model of gene regulation was established in prokaryotes, many laboratories have established that in eukaryotes, from simple yeast to mammals, the core principles are conserved at a mechanistic level. Of course there are also some important differences, and the one that is perhaps most obvious is that genes in bacteria are much more ‘Watson-Crick’ like, than they are in the nucleus of a eukaryotic cell, where they are compacted in an organised superstructure called the nucleosome, facilitated by a highly conserved family of histone proteins. These nucleosomes are then organised further into chromosomes; with which we are all undoubtedly very familiar. The organisation of the significantly larger genomes that is characteristic of eukaryotes, is one of the major differences between prokaryotes and eukaryotic cells.
As the DNA methylation community considered the role of DNA methylation and DNA methyltransferase(s), together with the complementary role of the methyl-CpG-binding domain proteins (MBDs), a burning issue presented itself. How could a system with such broad specificity explain the complexity of programmed gene expression during development?
Surveying the structure of gene expression
Biochemists have had a longstanding interest in chromatin, and classical biochemistry relied heavily on plant and animal tissues for their experimental materials: microbial biochemistry was largely confined to yeast, for obvious reasons! The focus on bacterial genes and their regulation was very much the preserve of microbiologists and geneticists: there was just too little material to be obtained from small flask cultures to match the appetite of the analytical techniques prevalent in the first half of the last century.

However, through a combination of their relative high abundance, their atypically low molecular weight, their nuclear localisation and of course and their basic character, histones (seen above packaging chromosomal DNA) became an attractive class of proteins for biochemists to explore. Histones were amongst those proteins that underpinned the development of protein chemistry methods and helped establish the fundamentals of protein primary structure. And this is where the second connection emerges between epigenetics and molecular evolution. It became clear very early in the history of protein chemistry that histones were amongst the most conserved classes of protein found in Nature
Unsurprisingly, all histones were rich in Lys and Arg residues, but it was the identification of modifications to the basic amino acids, lysine and arginine in 1960s by Ken Murray (working as a post-doc at Stanford, and who would later receive a knighthood as one of the UK’s most successful “translational” molecular biologists) and Alfred Mirsky at the Rockefeller, that would prove critical in the field. Their meticulous analysis of the chemical structure of histone primary structure would ultimately provide the fuel needed to inspire further developments in epigenetics research, via the regulated modification of histones.
Let’s take stock for a minute. By the end of the last century it was clear that all cellular genomes were composed of DNA. In many organisms, the four core bases of DNA (the pyrimidines denoted by C and T, and the purines G and A) are supplemented by modified forms, including 5-methyl C. Eukaryotes, unlike prokaryotes, package their DNA in the form of chromosomes with the aid of a group of highly conserved, small basic proteins called histones. Furthermore, histones may be post-translationally modified, commonly on the side chains of the basic amino acids; lysine and arginine. These modifications to DNA and histones have formed the focus for much of the mechanistic epigenetics work published over the last 25 years in particular.
The article by Mukherjee, sets out to celebrate the idea that these two phenomena: DNA methylation and/or histone modification, are at the heart of the orchestrated programming of gene expression during organismal development. It remains difficult however, as Ptashne (and others) have pointed out, to reconcile the evolutionary inconsistencies in this proposal. For example, one of the geneticists’ favourite model organisms Drosophila melanogaster manages just fine without C-5, cytosine methylation. And aren’t histones, masters of sequence-independent DNA recognition? And isn’t nucleotide sequence-specificity an indispensable part of genetic programming?
Genotype vs phenotype
 Reading the first edition of Conrad Waddington’s landmark text (1939) recently, I very much enjoyed his discussion of epigenetics (pp154-157).
Reading the first edition of Conrad Waddington’s landmark text (1939) recently, I very much enjoyed his discussion of epigenetics (pp154-157).
“The concepts of genotype and phenotype are defined in the first place in relation to differences between whole organisms. They are not adequate or appropriate for the consideration of the development of the differences within a single organism. In this connection we do, indeed require the concept of the hereditary (chromosomal) constitution of the zygote, and we can without danger extend the meaning of the word genotype to cover this. But the difference between an eye and a nose, for instance, is clearly neither genotypic nor phenotypic. It is due, as we have seen, to the different sets of developmental processes which have occurred in the two masses of tissue; and these again can be traced back to local interactions between the various genes of the genotype and the already differentiated regions of the cytoplasm in the egg. One might say that the set of organizers and organizing relations to which a certain piece of tissue will be subject during development make up its “epigenetic constitution” or “epigenotype”; then the appearance of a particular organ is the product of the genotype and the epigenotype, reacting with the external environment. In transplantation experiments, such as those described above, it is the epigenotype which is altered.”
It seems to me that Waddington uses the term epigenetics to contrast the plasticity of ‘phenotype’, with the ‘constancy’ of genotype, in the context of embryology and development. Waddington is clearly providing a prescient vision of systems biology. I think by drawing on Waddington’s use of the term epigenetics in his discussion of phenotypic change during the lifetime of an organism, and linking it directly with the subsequent discoveries in genome and histone modification Mukherjee has created some confusion. Something that must be avoided in the education of scientific novices and when informing the public about contemporary scientific discoveries.
Future directions
 The global investment in molecular epigenetic research and the many publications, reviews and books are beginning to suggest that diet (for example) can elicit epigenetic ‘commands’ that override transcription factor mediated regulation. The question for me is whether these ideas are experimentally grounded and sufficiently robust to challenge the endurance of the Jacob and Monod model. It remains unclear how significant a role nucleotide and histone modifications have on organismal development. However, it is absolutely clear that pharmacological perturbations of the enzymes of nucleic acid and histone maturation and the selective ablation of the encoding functions, can wreak havoc in model organisms, such as mice. Only through further research will a clear picture emerge of the confluence of nucleic acid and histone modification in the grand scheme of things.
The global investment in molecular epigenetic research and the many publications, reviews and books are beginning to suggest that diet (for example) can elicit epigenetic ‘commands’ that override transcription factor mediated regulation. The question for me is whether these ideas are experimentally grounded and sufficiently robust to challenge the endurance of the Jacob and Monod model. It remains unclear how significant a role nucleotide and histone modifications have on organismal development. However, it is absolutely clear that pharmacological perturbations of the enzymes of nucleic acid and histone maturation and the selective ablation of the encoding functions, can wreak havoc in model organisms, such as mice. Only through further research will a clear picture emerge of the confluence of nucleic acid and histone modification in the grand scheme of things.
I would encourage readers to take a look at the primary sources, including the elegant mathematical analysis of Turing, in order to ensure there is an appropriate level of balance in the selection of evidence when presenting scientific research to budding scientists and the wider public. Our understanding of the fundamental principles of gene regulation is also a matter of life and death!
Further reading:
- General properties and roles of histones
- The DNA methylome
- Histone Deacetylase (HDAC) inhibitors and therapeutics
- Cytosine modification discovery by Hotchkiss (1948)
- Restriction and Modification discoveries (1952 and 1953)
- Histone modification (Murray and Mirsky)
- Early C5 DNA methyltransferase sequences: Bestor and Trautner
- Methylcytosine binding proteins from Bird’s work
- Science special edition on epigenetics
- PNAS core concepts: epigenetics
- Ptashne on epigenetics
This post is the first in our epigenetics series. If you are interested in reading more on this topic, you can also check out the October issue of The Biochemist magazine on the theme of epigenetics.


3 thoughts on “Epigenetics: past and present”