By Emma Pettengale, Portland Press
 You might have seen the recent news story about Judy Perkins – the woman cured of terminal breast cancer using her own immune cells in a world first. Some doctors believe that the pioneering development could mark a “paradigm shift” in cancer research and we’re going to take a look at the case, and explain the science behind it!
You might have seen the recent news story about Judy Perkins – the woman cured of terminal breast cancer using her own immune cells in a world first. Some doctors believe that the pioneering development could mark a “paradigm shift” in cancer research and we’re going to take a look at the case, and explain the science behind it!
Tumour-infiltrating lymphocytes (TILs) are a type of white blood cell thought to represent the immune response directed against the antigens expressed on tumour cells – you can think of them as your cancer killing cells. Go team!
An important part of the immune system is its ability to distinguish between the normal cells in your body and those it sees as foreign or dangerous, which it does by recognising ‘antigens’. In most cases, the mutations that cause a cell to become cancerous also trigger the immune system to destroy it. However, cancer cells sometimes find ways to avoid being attacked by the immune system and become a tumour. Immunotherapy drugs aim to increase or improve the immune response to cancer cells and drugs such as Keytruda are already widely used, however complete responses are infrequent with current immunotherapies.
Certain proteins made by some types of immune system cells help keep immune responses in check and can keep your immune system from killing cancer cells. When these protein checkpoints are blocked, a previously exhausted immune system is converted into an effective response capable of killing the cancer cells. Checkpoint inhibitors are drugs that target these checkpoints and they hold a lot of promise as cancer treatments.
Garth W. Tormoen, Marka R. Crittenden, Michael J. Gough
Emerging Topics in Life Sciences 2017, 1 (5) 493-500; DOI: 10.1042/ETLS20170066
www.emergtoplifesci.org/content/1/5/493
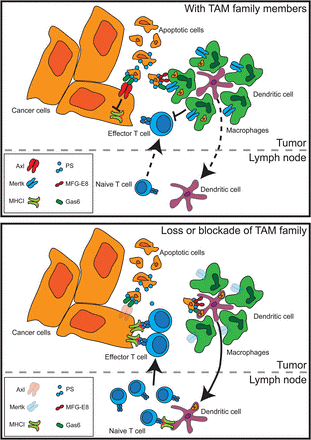
Effect of TAM family members on the immune response to cancer cell death.
Activation of Axl or Mertk on cancer cell results in suppression of antigen processing and presentation to T cells, while Mertk on tumour macrophages results in phagocytosis of dying cells to the exclusion of dendritic cell uptake. Cell uptake drives suppressive differentiation of macrophages, which can, in turn, suppress T cells in the tumour environment. Where TAM family members are absent, antigen is more available for uptake by dendritic cells that can expand naive T cells that can traffic to the tumour. Tumour-infiltrating T cells find decreased macrophage suppression and increased antigen presentation, permitting increased control of tumours.
Back to Judy Perkins. Aged 52 and from Florida, Judy’s case was terminal, before she underwent an experimental new type of immunotherapy and in a world first, has seen a “complete and durable regression” of her advanced breast cancer. This new approach combined two different forms of existing immunotherapy. To begin with, different therapies target different mutations, so the research team took a sample of Perkins’ tumours and sequenced her cancer’s DNA. They found 62 mutations unique to her cancer, including four that can be targeting by white blood cells. TILs specially targeting these mutations were removed and multiplied in the lab, with 90 billion eventually injected back, in conjunction with checkpoint inhibitors.
Peter Johnson, Professor of Medical Oncology at Southampton General Hospital, said: “This is another piece of evidence confirming that some cancers are recognisable by the body’s immune system and that if this can be stimulated in the right way, even cancers that have spread to different parts of the body may be treatable.”
According to Tom Misteli, from the National Cancer Institute, “This is an illustrative case report that highlights, once again, the power of immunotherapy.”
In our recent issue of Emerging Topics in Life Sciences, Francesco Marincola from AbbVie Immune Oncology Discovery, described how the second generation of cancer immunotherapy aims to go beyond checkpoint inhibitors and overcome cancer immune resistance. His introduction to the cancer immunology themed issue is available without a subscription.
In another recent trial, 40% of men given just months or weeks to live after being diagnosed with prostate cancer survived for more than a year. Pembrolizumab is another checkpoint inhibitor, and a trial led by a team at the Institute of Cancer Research (ICR) saw the astonishing results, presented in June at the American Society of Clinical Oncology annual meeting.
Professor Johann de Bono from the ICR said “I have these men who are basically dying, with weeks to months to live, who we gave this drug to and had complete responses. Their cancers shrunk, disappeared actually, with minimal cancer left on scans. These are amazing results, and these are men whose cancers had all the treatments, they had everything possible, they’ve got no treatments left, and they are in trouble. They have very short life spans left.”
If you’d like to find out more about recent clinical trials on cancer immunotherapies, the results of two phase III clinical trials – KEYNOTE 040 (pembrolizumab) and CheckMate 141 (nivolumab) are critically examined by Sara I. Pai and Lori J. Wirth from Harvard University in this recent review. In both trials, host immune responses result in an overall improved clinical prognosis and there is evidence that biomarkers may help to identify patients most likely to derive clinical benefit from immune checkpoint blockade.
Dr Steven Rosenberg, from the National Cancer Institute in Bethesda, Maryland leads the team who treated Judy, he said “Because this new approach to immunotherapy is dependent on mutations, not on cancer type, it is, in a sense a blueprint we can use for the treatment of many types of cancer.”
So while chemotherapy poisons the body and radiotherapy irradiates cells indiscriminately, immunotherapy takes the precise nature of our immune system and specifically magnifies the cancer killing cells, allowing them to kill tumours.
Further reading
- Emerging Topics in Life Sciences: Cancer Immune Resistance issue (2017) www.emergtoplifesci.org/content/1/5
- Immune recognition of somatic mutations leading to complete durable regression in metastatic breast cancer, Nature Medicine 24, 724–730 (2018) www.nature.com/articles/s41591-018-0040-8
- BBC News, ‘Remarkable’ therapy beats terminal breast cancer, www.bbc.co.uk/news/health-44338276
- Sky News, Advanced breast cancer ‘eradicated’ in world first news.sky.com/story/advanced-breast-cancer-eradicated-in-world-first-11395089
- Sky News, Immunotherapy prostate cancer drug gives ‘amazing results’ news.sky.com/story/immunotherapy-could-benefit-prostate-cancer-patients-11393083

