By Chloé Yeung, Institute of Sports Medicine Copenhagen, Denmark
At the start of October, the 2017 Nobel Prize in Physiology or Medicine was awarded jointly to Jeffrey C. Hall, Michael Rosbash and Michael W. Young for their discoveries of molecular mechanisms controlling the circadian rhythm. In recent years, the very same molecular mechanisms have been discovered in our peripheral tissues and we’re now beginning to understand why circadian rhythms are also needed in our musculoskeletal tissues including muscle, cartilage and tendon.
The circadian clock is an evolutionarily conserved system that life forms have developed to allow appropriate physiology and behaviour to be coordinated with the environment. It helps our bodies to anticipate and prepare for, rather than just adapt to, the changes that occur in a 24-hour day. Tissues with endogenous 24-hour rhythms are known as peripheral clocks and it is now believed the purpose is to regulate tissue-specific biological processes.
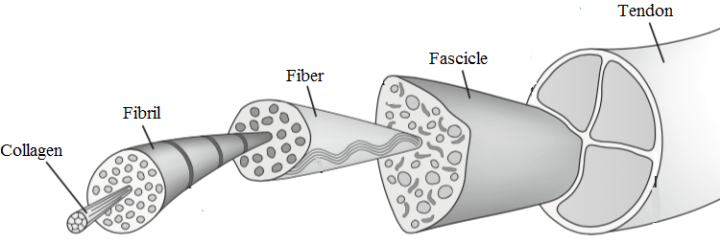
Tendons connect and transmit forces from muscle to bone, enabling us to go about our daily activities that range from gripping a toothbrush or cup of tea to getting out of bed and walking around. A tendon’s ability to undergo repeated cycles of mechanical loading is owed to its extracellular matrix or ‘matrix’ of parallel bundles of fibrillar collagen, which makes up for 75% of the dry tissue mass.
A process known as matrix homeostasis regulates the amount, composition and organisation of matrix proteins by balancing addition of new proteins and removal of damaged proteins, to help maintain healthy tendons. For unknown reasons matrix homeostasis is sometimes not enough to restore the tendon matrix, e.g. after injuries and as we age. Tendinopathies can then develop, which are associated with chronic pain, inflammation and disability, and are the second most common orthopaedic condition behind osteoarthritis.

Tendon tissue is a peripheral circadian clock that controls the 24-hourly rhythmic expression of over 700 genes. Disruption of core circadian genes in mice causes them to lose both their gene expression and behavioural rhythms. We found that tendons from mice with disrupted clock genes exhibited ectopic calcification, were thickened and were mechanically weaker, supporting the idea that a robust 24-hour rhythm is important for healthy, functional tendons.
Decline of circadian rhythms in peripheral tissues, including cartilage and tendon, has been observed in aged mice, which could help explain the high prevalence (1-in-4 people over the age of 40) of chronic musculoskeletal disorders in the older population. However, it is unknown why peripheral circadian rhythms decline with age when the central oscillator does not. In addition, it is still not clear why tendon and other peripheral tissues require a 24-hour expression pattern of certain genes. Possible reasons could be: to synchronise particular biological processes with the rest-activity cycle, or with other pathways in order to coordinate a cascade of events; to temporally compartmentalise biological processes that may interfere with each other; or to maintain constant levels of signalling molecules or enzymes by gating their expression. Further understanding of the mechanisms and the reasons why peripheral circadian rhythms dampen with age would make peripheral clocks great novel therapeutic targets in the prevention and management of tendinopathies and other age-related musculoskeletal disorders.
About me
 I am a postdoctoral research fellow at the Institute of Sports Medicine Copenhagen, Bispebjerg Hospital in Denmark. I did my PhD at the Wellcome Trust Centre for Cell-Matrix Research, University of Manchester. My research focus is to understand how circadian clocks of the musculoskeletal system regulate tissue homeostasis and how they age.
I am a postdoctoral research fellow at the Institute of Sports Medicine Copenhagen, Bispebjerg Hospital in Denmark. I did my PhD at the Wellcome Trust Centre for Cell-Matrix Research, University of Manchester. My research focus is to understand how circadian clocks of the musculoskeletal system regulate tissue homeostasis and how they age.
You can find me on Twitter @ccstorytime and on ResearchGate and I also have my own blog Stories of Chloé, where I write about my experience of moving abroad to pursue an independent research career, and about what my friends have done since their PhDs and their advice to those who are considering doing a PhD.


One thought on “Why can tendons tell time?”