By Emily May Armstrong, University of Glasgow
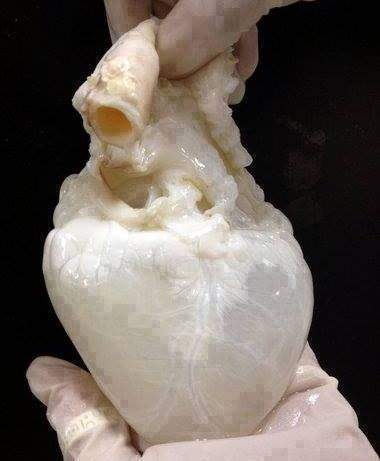
There is currently an imbalance between the number of organs that are donated and the needs of the population – there just aren’t enough organs to go round for those that need them. Tissue regeneration and the building of artificial organs could be a way to combat this. Currently, there’s a massive drive toward improving, creating, and manufacturing cheap and safe alternatives to traditional, mammalian-based biomaterials. Biomaterials take many forms: solid inert scaffolds, decellularized ghost organs, hydrogels, pastes, and powders. These materials can be passive, actively deliver drugs or tissue growth promoting materials, like hydroxy-apatite releasing hip implants which promote bone ingrowth. This is where plants come in, our photosynthetic friends are diverse, adaptable, and cheap to grow – making them perfect candidates for novel biomaterial production.
Encouraging growth
Having a bone implant such as a hip replacement is a serious operation: an infection or rejection can be highly problematic. Titanium is the gold-standard for nearly all implants; it is strong, stress resistant and rigid. The titanium-tissue interface must encourage ‘osseointegration’, where the introduced biomaterial must successfully integrate into the surrounding bone. Implant coatings facilitate this, encouraging bone re-growth and preventing rejection responses. A plant cell wall is comprised of pectins, complex sugars which provide stability and pathogen barriers in planta. A study by Kokkenen and colleagues treated apples with enzymes, which separated sugar sub-groups from their main carbon backbone. These were applied at nanometre thickness to titanium dental implants. They showed apple pectin coated titanium implants facilitated osseointegration with reduced rejection responses in rat bone tissue culture.
Engineering scaffolding
Biomaterials are being used as ‘scaffolds’ for both tissue engineering and cellular manufacturing. Scaffolds must be inert, allow fluid transport, and support basic cell functions. Even with 3D printing, distilling the highly complex needs of a biomimetic material into an affordable and functional product is difficult. Plant vasculature tissues offer an attracting scaffolding alternative, as their structure and function mimic human tissues.
Decellularization is a process where native cells are removed from tissues using detergents and bleach, cleaning up any plant DNA or protein. The resulting decellularized tissues are lacking in pigment, and most of the plants tested in Fontana and colleague’s 2017 paper retained their ultrastructure. Some decellularized plant tissues could carry up to 40 times more water than their ‘natural’ counterparts. The team coated the tissues with conjugated adhesive proteins, and demonstrated that when these proteins attached to decellularized plant tissues, they allowed adhesion of human skin cells. The cells continued to grow for 50 days, and grew larger than they usually do when grown in standard conditions. The cells also adhered to the micro- and macrostructures of the plant tissues, and were responsive to topographical cues, which can guide cell behaviour.
Cell-derived cellulose
Cellulose is the most abundant biopolymer on earth. Like pectin, it provides structural integrity to plant cell walls, and provides an appealing target for biomaterial exploitation. As it can be biochemically modified with ease, Andrew Pelling’s group from the University of Ottowa turned (again), to apples. They developed a cheap and simple way to extract cellulose from the apple hypanthium (the crunchy bit we eat), and implanted it in mice.
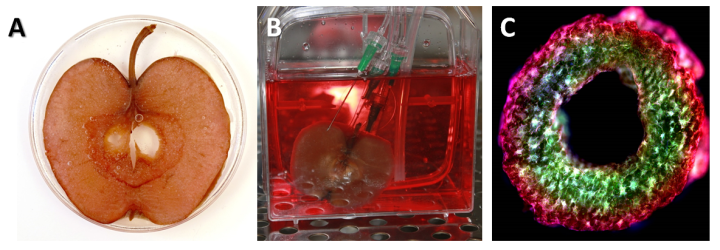
The Pelling Group’s apple-cellulose scaffolds. A) shows a decellularized apple cross section which has been recolonised by human cells which adhere to the remaining cellulose. B) shows the cell seeding process around the apple scaffold, and C) shows an ultra-high-resolution cross section of an apple cellulose ring structure. Images provided and taken by Andrew Pelling from the University of Ottowa.
The hypanthium is a perfect biomaterial chassis, as it carries water and nutrients throughout the tissue. The tissue was decellularized, leaving behind an intact extracellular matrix, allowing for native cell re-colonisation. They reported minimal immune responses and few severe infections, and no deaths resulting from implantation – an improvement compared to standard mammalian scaffolds. Furthermore, in vitro studies showed mouse fibroblasts, myoblasts and human HeLa cells were able to colonise the cellulose scaffolding, making it a potential game changer in the world of biomaterial implantation. In their latest paper, they take this technology even further by using cellulose scaffolds to rapidly prototype complex tissue structures in situ, and are beginning human implantation trials this year.
Spinach-human interfaces
Taking this research one step further is the Gaudette group from the Worcester Polytechnic Institute, bringing decellularization to almost full functionalisation in their Biomaterials paper ‘Crossing Kingdoms: Using decellularized plants as perfusable tissue engineering scaffolds’. Noticing a gap in the biomaterial market, they took advantage of plant vasculature tissue, which mimics mammalian vasculature. The central xylem and phloem allow for transport of water and nutrients to distant cells, much like blood vessels in humans. They decellularized a spinach leaf (obtained from their local market), and tested the leaf with a red dye, which showed minimal liquid leakage.
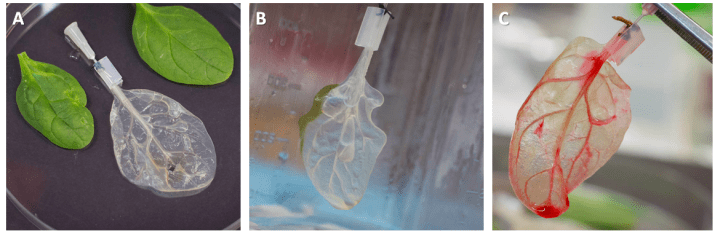
The Gaudette Group’s decellularized spinach leaf-heart. A) hows a decellularized spinach leaf compared to its intact counterparts, B) shows the catheter apparatus used to perfuse the tissue and C) shows red dye perfused through spinach scaffold recellularised with cardiomyocytes. Images provided by The Gaudette Lab from the Worcester Polytechnic Institute.
For any engineered leaf-heart to work, the smallest vessels must carry red blood cells, which are 6-8µm in diameter. This was confirmed using fluorescent beads, and showed these vessels could carry these vital cell types. Content with this proof of concept, they moved on to recellularization with human cells. Human endothelial cells were introduced inside the vasculature lumen, whereas mesenchymal stem cells were seeded onto the outer tissue. These readily adhered and grew. Taking this even further, cardiac muscle cells (cardiomyocytes) were seeded onto the surface of the leaf scaffolds. Cardiomyocytes have the capacity to contract, or ‘beat’, on their own, which is key to heart function. Amazingly, the seeded cardiomyocytes on the spinach could grow, and beat on their own.
There’s a lot to be said about the power plants have over our scientific imagination. From providing us with dinner, to providing us with ground-breaking medical applications, plants continue to be at the forefront of exciting, engaging and applicable research.
 About me
About me
I am a PhD Student at the University of Glasgow, studying epigenetic regulation of gene transcription in plant roots under salinity stress.
You can find more out about me and my disability outreach work on my website. You can also contact me on Twitter @emilyXarmstrong, or keep up to date with my daily lab life on my science Instagram @theradicalbotanical.
This post is the first in our biomaterials series. If you are interested in reading more on this topic, you can also check out the February issue of The Biochemist magazine on the theme of biomaterials.


Super geschriebener und informativer Artikel :-). Eine sehr gute Aufstellung. In diesen Blog werde ich mich noch richtig einlesen
LikeLike